While many changes are “under the hood”, I wanted to describe some of the more significant updates that have been added recently.
Organization Perhaps the most important update is a change in the organization of the buttons and checkboxes that allow you to animate the symmetry operations and display the symmetry elements of a particular molecule. In previous versions, they were labeled in a way that did not carefully distinguish between symmetry elements and symmetry operations.
The controls are now labeled consistently by operation, with a drop-down menu to select the grouping of operations. The default grouping is by point group class (see Figure 1 below). Other options include grouping by symmetry element or no grouping at all. The grouping by element can be useful to highlight point groups that are chiral (pure rotational groups).

Figure 1: Symmetry operations for a C3v molecule, with operations grouped by point group class
The reorganization of controls also moves the element display checkbox over to the right and only displays one checkbox if multiple operations occur about a single element. Finally, the up arrow symbol now represents the reorient control and will align the molecule to look down the respective axis or across a plane of symmetry.
Symmetry Analysis The other major change is the addition of the Symmetry Info tab on the right side panel of the Gallery. At the top is a table showing sets of symmetry equivalent atoms, followed by the framework group1 and the respective vibrational representation.
Just below the table of symmetry equivalent atoms is a Color by: Symmetry control which will color the atoms in the molecule by symmetry equivalence, giving a unique color to each set of symmetry equivalent atoms. Clicking the CPK button will return the atoms to their original colors. An example molecule is shown in Figure 2. This view can be clear demonstration that atoms of the same element are not always related by symmetry. On a more technical note, all the symmetry information presented is generated dynamically using the SYVA symmetry analysis program2, so future updates may include additional analysis.
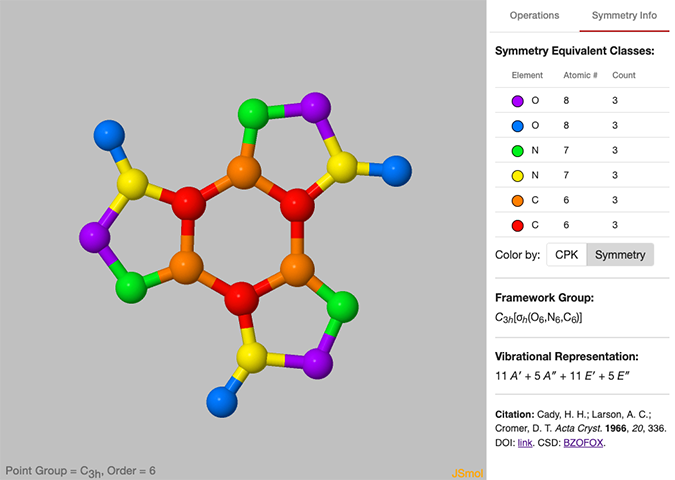
Figure 2: Symmetry Information for the benzotrifuroxan molecule, with atoms colored by symmetry equivalency.
If available, the citation for the given structure, including a link to the original journal article (DOI) and the associated structure in the Cambridge Structural Database (CSD) is also given.
Interface Enhancements One obvious addition is the (return of the) slider. When operations are animated, the slider will move along with the animation. Clicking and dragging the slider will step through the most recent animation.
One other recent addition is the ability to export animations as GIFs. If you hold down the SHIFT key while clicking on an operation button, a popup will ask for a filename. The animation will proceed (very slowly) and when complete you will be given the option to download the corresponding GIF. Warning - it is advisable to resize your window such that the molecule portion is not too large. The process is computationally intensive and can potentially generate very large files. This feature is still very experimental.
New Molecules Finally, the collection of molecules now numbers over 120! Additions since the last major update include: C70 (D5h), chloromethane (C3v), benzene-1,3,5-triol (C3h), tris(ethylenediamine)cobalt(III) (D3), sulfur tetrafluoride (C2v), cyclobutadiene dianion (D4h), naphthalene (D2h), diborane (D2h), pentamantane (Td), boron trifluoride (D3h), and 1-bromo-1-chloroethane (C1), among many others.
Thanks as always for the feedback and suggestions.